- 阅读
- 阅读
- 技巧
- 百科
- 试题
- 文化
- 技能
世界卫生组织总干事谭德塞近日表示,世卫组织一个小组正在中国与中国科兴公司和中国国药集团开展合作,评估其生产的新冠疫苗。
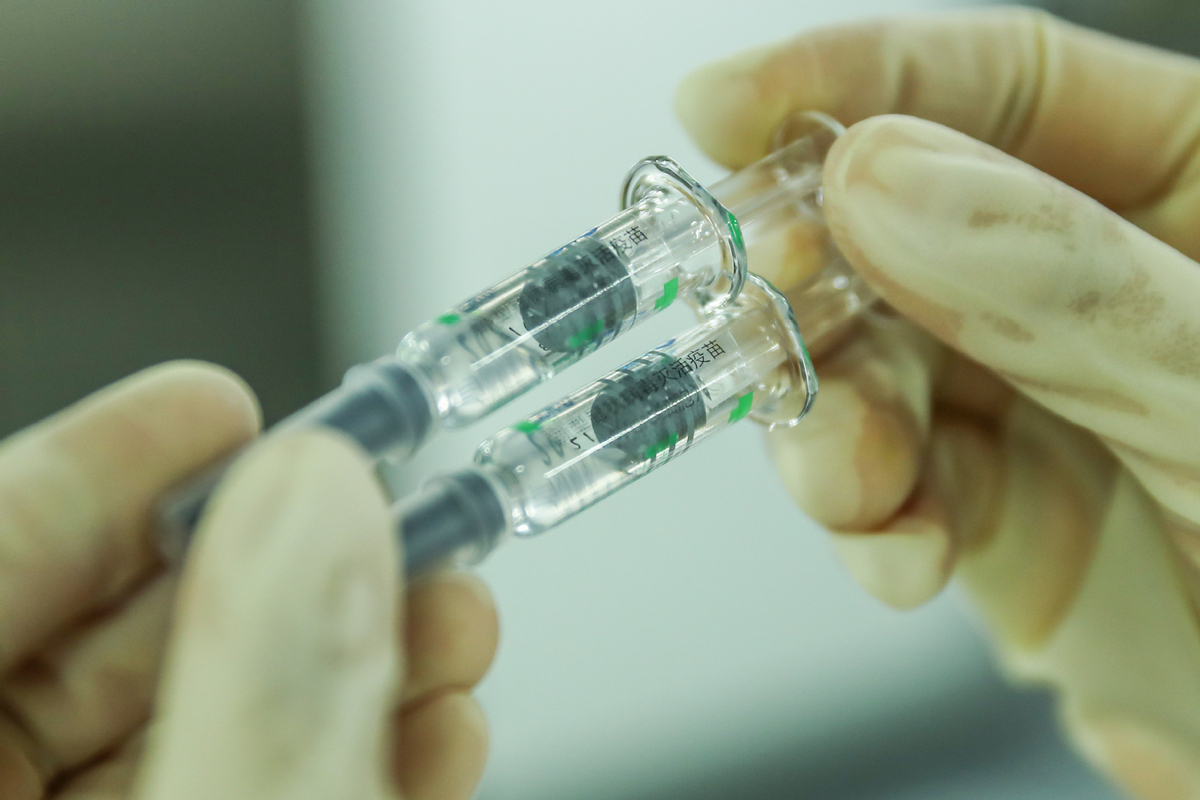
"I'm pleased that a WHO team is in China currently working with
producers of the Sinovac and Sinopharm vaccines to assess compliance
with international quality manufacturing practices ahead of potential
emergency use listing by WHO," Tedros said.
谭德赛表示:“世卫组织一个小组正在中国与中国科兴公司和中国国药集团开展合作,评估这些企业在国际品质生产规范方面的合规情况,以便世卫组织未来将这些企业生产的疫苗列入紧急使用清单,很高兴看到这一进展。”
He noted that WHO continues to ask vaccine manufacturers from
around the world to move swiftly to provide necessary data that will
allow the organization to consider them for emergency use listings.
谭德塞呼吁全球疫苗生产商提供必要数据,以便世卫组织在审核后将相关疫苗列入紧急使用清单。
"While we are hopeful about the safe and effective vaccines that
are being rolled out, we want to see this sped up and vaccines allocated
equitably in the coming weeks," Tedros said.
谭德塞表示:“我们对推出安全有效的疫苗充满希望,同时我们也希望看到这一进程加快,并在未来几周内公平分配疫苗。”
国产疫苗开始接种
More than 9 million doses of homegrown coronavirus vaccine have been
administered in China, the National Health Commission said on Saturday.
国家卫健委1月9日表示,我国已累计开展新冠病毒疫苗接种900多万剂次。
A number of countries, including Egypt, the UAE, Jordan and
Indonesia, have authorized the COVID-19 vaccines produced by China for
emergency use. And many more countries, including Brazil, Chile,
Malaysia, the Philippines, Thailand and Nigeria, have ordered Chinese
vaccines or are cooperating with China in procuring or rolling out the
vaccines.
埃及、阿联酋、约旦以及印度尼西亚等国已经授权中国新冠疫苗在本国紧急使用。巴西、智利、马来西亚、菲律宾、泰国以及尼日利亚等国已经订购中国疫苗,或正在与中国就疫苗采购或推出进行合作。
【相关词汇】
药品和疫苗研发
drug and vaccine development
免疫系统
immune system
有效性和安全性研究
safety and efficacy research
人类卫生健康共同体
a global community of health for all
疫苗联合研发
joint research and development of vaccines
来源:英语点津
参与评论